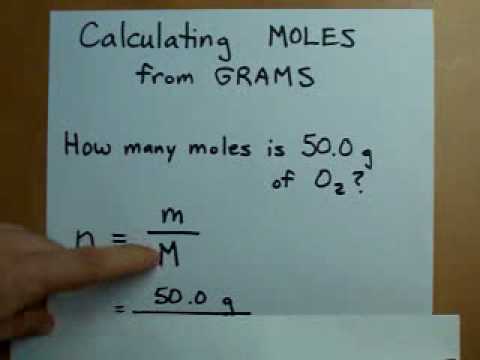 
The moles to atoms formula: 1 Mole 6.0221415E+23 atoms The moles to atoms conversion and atoms to moles conversion are interchangeable to each other. We take C-12 as a standard of measuring Avogadro’s Number. N = m / M and The mass m in grammes divided by the molar mass M in g/mol equals the quantity of material n in moles. The value of the moles is equal to the number of atoms of 12 g C-12 Carbon atoms 6.022140857 x 1023 atoms. 
worksheet review packet with more combined mole calculations worksheet answers, below we and. Now that you know the substance's molar mass, you can use the total mass in grammes to compute the quantity in moles. How many atoms of tin are found in 3.50 moles of tin 10. Of course, if you wouldn't have the molar mass and would like to bypass this step, you could use our molar mass calculator to quickly determine a substance's molecular mass. Because each molecule has two hydrogen atoms and one oxygen atom, the formula should be:Īs a result, H 2O has a molar mass of 18.01528. Molar concentration (also called molarity) is the number of molecules of a substance, expressed in mole units (see above), present in a certain volume of a. Hydrogen has an atomic mass of 1.00794 and oxygen has an atomic mass of 15.9994. Two parts hydrogen (H) and one component oxygen (O) make up H 2O. On the periodic table, you can find the atomic mass.Įxample: Let's look up the molar mass of water, for instance (H 2O). G/mol is a common unit of measurement for molar mass.Ĭalculate molar mass by first determining the number of atoms in the molecular formula, then multiplying the atomic masses of each element to obtain the molecular weight. The molar mass of the chemical equation is the first step in determining the amount of moles of a substance. We'll show you how to convert mass to moles in only a few steps if you follow along. You could be asking how to figure out how many moles a substance has based on its mass, volume, or quantity. Because the mole is a unit of measurement, every material has the same number of atoms, molecules, and other particles in one mole. Formula: Mole Atom 6.0221415E+23 Atom Mole / 6.0221415E+23 where, Atom Number of atoms 1 Mole 6.0221415E+23 Atom people found this article helpful.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
